A Comprehensive Investment Analysis of AI Drug Discovery (AIDD): Welcoming the "GPT-2 Moment"
$Insilico Medicine (03696)$ $QuantumPharm (02228)$ Recursion Pharmaceuticals $ Schrodinger
I recently listened to a podcast with Anthropic CEO Dario Amodei. When asked about the next industry AI will disrupt, he pointed to biopharma. Anthropic also recently acquired a biotech company for $400 million. This made me curious about the actual impact of AI on drug R&D and which companies stand to benefit. I found it quite fascinating and had Gemini summarize the key takeaways:
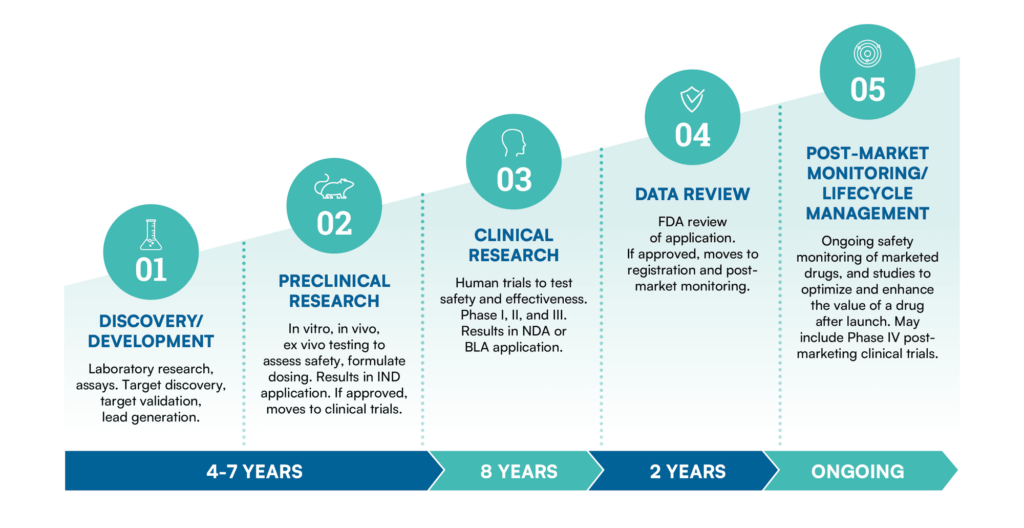
I. Macro Catalysts: Traditional Pharma Faces Patent Cliffs and Efficiency Bottlenecks The global biopharmaceutical industry is at a critical crossroads. The traditional drug R&D model has become extremely expensive and inefficient, typically requiring up to 10 years and billions of dollars in investment, with clinical failure rates hitting 90% in some crowded target areas.
More urgently, multinational pharmaceutical companies (MNCs) are facing a massive "patent cliff." It is estimated that by 2035, pharma giants will face up to $115 billion in revenue loss due to the loss of exclusivity (LOE), creating a pipeline revenue gap of approximately $40 billion before 2030. To rescue their tumbling single-digit return on investment, the entire industry is being forced to turn to AI and technological solutions to lower R&D costs and replenish their innovative product pipelines.
II. The "GPT-2 Moment" for Biotech: Initial Clinical Validation
Human genes and cells are highly programmable, with underlying logic strikingly similar to the 0s and 1s of computer programming. Thanks to AI, drug discovery is experiencing the technological dividends of the internet age on a massive scale for the first time. Currently, the field is in the "GPT-2 stage" of its technology maturity curve: we have witnessed clear clinical proofs-of-concept (PoC), but we might still be over two years away from the first fully AI-discovered and designed drug receiving market approval.
As of the end of 2024, more than 75 AI-derived molecules had entered human clinical trials. Early data shows that the success rate of AI-designed molecules in Phase I clinical trials reaches 80%-90% (compared to a historical industry average of about 52%). For instance, Insilico Medicine's Rentosertib (ISM001-055)—a drug for idiopathic pulmonary fibrosis where the target was discovered and the molecule designed entirely by AI—published its positive Phase IIa clinical data in Nature Medicine in June 2025, providing a strong confidence boost for the entire industry. Meanwhile, TAK-279, designed by Schrödinger through a platform combining physics and machine learning, has successfully advanced into Phase III clinical trials.
III. Core Bottlenecks and Solutions: Data Acquisition and Automated Labs
The biggest constraint in this race is not computing power, but the difficulty of acquiring high-quality, proprietary biochemical data to train large models. Due to regulatory compliance and expensive experimental costs, breaking this data bottleneck typically involves two paths:
Federated and Synthetic Data: Partnering with governments or large pharma companies to unlock internal patient data (e.g., bypassing privacy restrictions via federated learning mechanisms). Concurrently, frontier companies are leveraging quantum physics to generate synthetic data or developing specialized biological foundation models. For example, Insilico Medicine's "MMAI Gym" model training framework can significantly boost target search success rates from 20% to roughly 70%.
Autonomous "Self-Driving" Labs: Using AI combined with automated robots to exponentially increase testing and development speed. This closed-loop "design-make-test-analyze" workflow not only reduces R&D costs but also endlessly generates proprietary data for AI to learn from, creating a highly defensible moat.
IV. Geopolitical Shift: China Becomes a Crucial Hub for AI Drug Discovery In the global investment landscape, Tech-Bio companies listed in China and Hong Kong are demonstrating extremely high investment value. Although the U.S. dominates foundational AI models, China has established advantages in applied pharmacology and clinical execution. By early 2026, China accounted for nearly 70% of global AI-driven drug discovery patent applications. This advantage stems from China's massive and centralized clinical patient resources, a dense CRO/CDMO ecosystem, and fast regulatory approval pathways (e.g., the NMPA's drastically shortened IND review cycle for innovative drugs allows Phase I clinical safety data to iterate much faster than in the U.S. and Europe).
V. Two Mainstream Business Models: AI-CRO vs. End-to-End Pipeline
As the industry matures, companies are heading down two distinct commercial paths:
AI-CRO (Infrastructure/Platform Service Model): Represented by QuantumPharm (XtalPi). These companies do not directly undertake high-risk clinical trials. Instead, they use AI and automated workstations to provide services to large pharma companies, earning upfront payments, milestone payments, and sales royalties. This model is less capital-intensive and easier to achieve profitability. In fact, XtalPi recorded 803 million RMB in revenue in 2025 (a 201% YoY increase) and achieved a net profit of 124 million RMB. Its AI4S (AI for Science) business is poised for a commercial explosion across industries (such as new materials, consumer health, etc.) in 2026-2027 [1, 1]. They notably helped Pfizer confirm the most stable crystal form of the oral COVID-19 drug Paxlovid in just 6 weeks, proving immense value in solving real-world engineering problems.
End-to-End Pipeline (Pipeline Innovator): Represented by Insilico Medicine and Recursion (which recently merged with Exscientia ). These companies develop AI software and also advance clinical trials themselves. The downside is that clinical trials consume vast amounts of capital, imposing a heavy burden on companies lacking cash flow; the upside is that once the accuracy of the AI model and the drug's efficacy are proven in the clinic, their assets can be out-licensed at extremely high premiums. Insilico Medicine has successfully advanced over 20 clinical or IND-stage projects and secured global licensing deals worth over $2 billion in total (including the USP1 inhibitor project licensed to Exelixis).
VI. Summary and Outlook
The current valuation of the AI drug discovery sector bakes in extremely high future expectations, requiring investors to continuously monitor fundamentals. Although the overall success rate of Phase II clinical trials (around 30%-40%) is still comparable to traditional methods—indicating that AI currently solves mostly early-stage discovery efficiency issues—the industry is approaching ultimate validation as Insilico's oral Rentosertib plans to initiate Phase III trials in the second half of 2026 [1, 1].
While it may take at least two more years for a true blockbuster drug to be approved, the flurry of multi-billion-dollar acquisitions and strategic partnerships by major pharma companies proves one thing: the industry highly recognizes that AI drug discovery is the inevitable future. Once these models cross the critical threshold in accuracy and efficacy, they will trigger unprecedented commercial demand and value.
Breaking Modality Boundaries (From "Small Molecules" to Peptides and RNA): Most market awareness currently focuses on AI-designed small molecules, but between 2025 and 2026, AI has achieved major breakthroughs in more complex molecular modalities. For example, the PepiX™ platform, based on AI generative design and automated synthesis, is now being used to tackle next-generation peptide drug R&D for metabolic diseases. Meanwhile, AI-driven RNA-targeted therapies (like the RTX-117 pipeline for rare diseases) have rapidly secured clinical approvals in both the U.S. and China. You can emphasize to readers that the Total Addressable Market (TAM) for AI drug discovery is expanding rapidly.
Regulatory "Green Lights" and Clarifying Rules: Clinical and regulatory risks have always deterred large-scale traditional capital from entering this sector. In January 2026, the U.S. FDA and the European EMA jointly issued the Guiding Principles of Good AI Practice in Drug Development, signaling that top global regulators are not only embracing AI but providing clear standards and frameworks for its safe and compliant application throughout the product lifecycle. In addition, China's NMPA continues to optimize regulations, further compressing clinical trial approval times to 20 days and establishing "green channels" for highly effective innovative drugs. This synchronized global regulatory optimization has largely cleared compliance hurdles for AI drug discovery firms.
VII. Q&A: Three Core Questions About the Future of AI Drug Discovery
Q1: With the continuous evolution and open-sourcing of foundational models like AlphaFold, will existing AI drug discovery companies be disrupted or replaced?
My view: No. The progress of open-source models will actually strengthen specialized companies that possess "data moats" and "experimental validation capabilities."
First, open-source models primarily solve the baseline problem of general structural prediction, but they face a severe "data scarcity" bottleneck. Public biological and chemical datasets are often insufficient for training highly accurate drug discovery models. In the AI drug discovery sector, the true core moat is proprietary experimental data generated internally—especially high-quality "negative data" (i.e., failed experimental data) produced by strictly controlled automated labs, which is crucial for eliminating AI hallucinations and improving model reliability.
Second, the advancement of open-source large models essentially provides a better "foundation" for specialized models, allowing AI drug discovery companies to focus on differentiation and precise fine-tuning. More importantly, no matter how perfect the algorithm's predicted molecular compound is, it must ultimately be synthesized and validated in the real physical world. This is exactly why pure software companies are vulnerable to disruption, while companies with robust automated robotic wet labs (a closed-loop of dry and wet experiments) possess deep irreplaceability. Open-source models provide a smarter "brain," but the industry still needs precise "hands" to validate the work.
Q2: Will the proliferation of AI technology weaken the value of the CRO (Contract Research Organization) industry?
My view: The impact is highly positive. Rather than weakening CROs, AI will act as a catalyst to stimulate massive outsourcing and R&D demand.
Because AI drastically shortens the cycles for early-stage target discovery and molecular design and lowers upfront trial-and-error costs, pharmaceutical companies can push more drug candidates into the clinical stage faster and at a lower cost. An explosion in the number of candidates at the front end of the R&D funnel will inevitably lead to a sharp increase in demand for downstream CRO clinical testing and validation.
Additionally, AI technology makes it more feasible to tackle rare diseases, complex tumors, and entirely new therapeutic mechanisms. R&D for these complex diseases requires highly specialized knowledge, driving more outsourcing demand toward CROs with deep therapeutic expertise. By 2026, CROs will no longer be simple "outsourcing vendors" but "strategic co-development partners" integrating AI technology, regulatory intelligence, and global operational capabilities. Faster R&D and lower costs will inevitably bring prosperity to the industry's overall demand.
Q3: How vast is the future scope for AI in the scientific field? Why are you particularly optimistic about QuantumPharm's (XtalPi) business layout and philosophy at this stage?
My view: The future space is by no means limited to biomedicine, but extends to the broader "AI for Science" (AI4S) landscape.
Pharmaceutical R&D is essentially the physical and chemical recombination of molecules and atoms. If an AI model and automation system can accurately predict and synthesize drug molecules, it can equally be applied to materials science, fine chemicals, new energy batteries, and other fields. I am personally very optimistic about XtalPi's strategy because they haven't restricted themselves to being just "an innovative drug company"; instead, they position themselves as an "automated R&D infrastructure based on quantum physics and AI."
Entering 2026, XtalPi has successfully expanded its platform empowerment into non-pharmaceutical sectors such as new materials, agricultural technology, consumer health, and chemicals. They have deployed hundreds of automated robotic experimental workstations globally, capable of executing high-throughput experiments 24/7, boosting data collection capacity by tens of times. This philosophy of "underlying physical computation + robotic automation generating proprietary data" gives them a commercial footprint and ceiling far beyond the single biopharma track, demonstrating remarkable anti-cyclical resilience and cross-industry expansion potential.